A long-lasting neural probe
Researchers develop implantable device that can record a collection of individual neurons over months
By Leah Burrows
January 26, 2024
Recording the activity of large populations of single neurons in the brain over long periods of time is crucial to further our understanding of neural circuits, to enable novel medical device-based therapies and, in the future, for brain–computer interfaces requiring high-resolution electrophysiological information.
But today there is a tradeoff between how much high-resolution information an implanted device can measure and how long it can maintain recording or stimulation performances. Rigid, silicon implants with many sensors, can collect a lot of information but can't stay in the body for very long. Flexible, smaller devices are less intrusive and can last longer in the brain but only provide a fraction of the available neural information.
Recently, an interdisciplinary team of researchers from the Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS), in collaboration with The University of Texas at Austin, MIT, and Axoft, Inc., developed a soft implantable device with dozens of sensors that can record single-neuron activity in the brain stably for months.
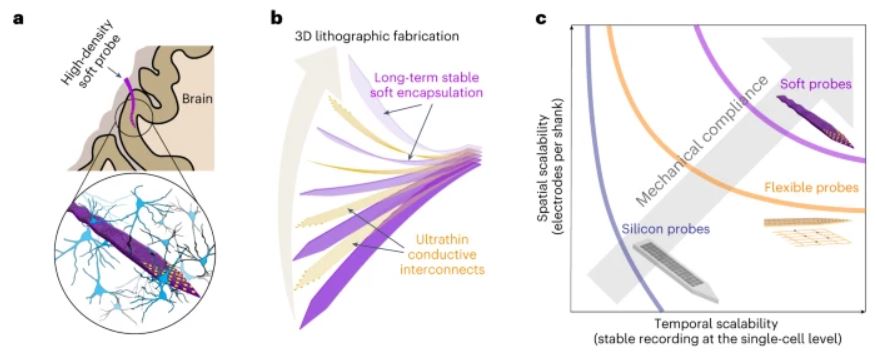
a. Schematic showing a spatiotemporally scalable neural probe implanted in the brain. The inset shows a 3D stacked microelectrode array encapsulated by soft elastomer layers, which can prevent ion permeation from the biofluid and maintain cell- and tissue-level mechanical properties, capable of providing a long-term stable interface with neurons at the single-cell level.
b. Schematic illustrating the 3D lithographic fabrication of multilayer thin-film soft encapsulation/conductive interconnects to scale up the number of electrodes integrated on the neural probe.
c. Spatial and temporal scalability of neural probes. Rigid silicon electronics can achieve high-density electrodes for neural recording but cannot stably record electrical activities from the same neurons over a long time period. Tissue-like flexible electronics can record electrical activities from the same neurons over a long time but do not offer scalability to increase the density and number of electrodes for neural recording. Soft electronics with a 3D electrode array can provide both spatial and temporal scalability for neural recording. (Credit: Nature)
The research was published in Nature Nanotechnology. ![]()
"We have developed brain–electronics interfaces with single-cell resolution that are more biologically compliant than traditional materials," said Paul Le Floch, first author of the paper and former graduate student in the lab of Jia Li, Assistant Professor of Bioengineering at SEAS. "This work has the potential to revolutionize the design of bioelectronics for neural recording and stimulation, and for brain–computer interfaces."
Le Floch is currently the CEO of Axoft, Inc, a company founded in 2021 by Le Floch, Liu, and Tianyang Ye, a former graduate student and postdoctoral fellow in the Park Group at Harvard.
To overcome the tradeoff between high-resolution data rate and longevity, the researchers turned to a group of materials known as fluorinated elastomers. Fluorinated materials, like Teflon, are resilient, stable in biofluids, have excellent long-term dielectric performance, and are compatible with standard microfabrication techniques.
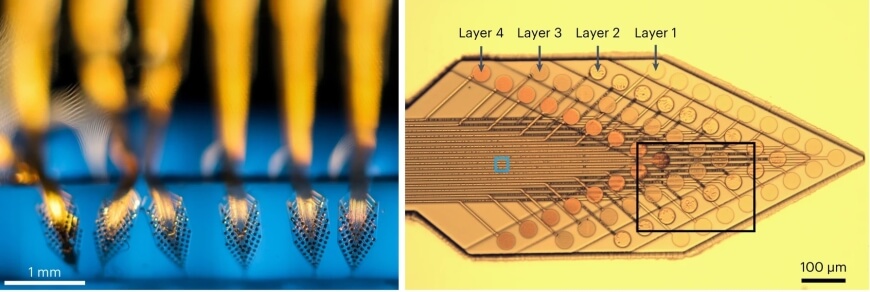
The researchers integrated these fluorinated dielectric elastomers with stacks of soft microelectrodes—64 sensors in total—to develop a long-lasting probe that is 10,000 times softer than conventional flexible probes made of materials engineering plastics, such as polyimide or parylene C.
The team demonstrated the device in vivo, recording neural information from the brain and spinal cords of mice over the course of several months.
"Our research highlights that, by carefully engineering various factors, it is feasible to design novel elastomers for long-term-stable neural interfaces," said Liu, who is the corresponding author of the paper. "This study could expand the range of design possibilities for neural interfaces."
The interdisciplinary research team also included SEAS Professors Katia Bertoldi, Boris Kozinsky and Zhigang Suo.
"Designing new neural probes and interfaces is a very interdisciplinary problem that requires expertise in biology, electrical engineering, materials science, and mechanical and chemical engineering," said Le Floch.
The research was co-authored by Siyuan Zhao, Ren Liu, Nicola Molinari, Eder Medina, Hao Shen, Zheliang Wang, Junsoo Kim, Hao Sheng, Sebastian Partarrieu, Wenbo Wang, Chanan Sessler, Guogao Zhang, Hyunsu Park, Xian Gong, Andrew Spencer, Jongha Lee, Tianyang Ye, Xin Tang, Xiao Wang and Nanshu Lu.
Harvard's Office of Technology Development has protected the intellectual property associated with this research and licensed the technology to Axoft for further development.
Authorship, funding, disclosures
The research was supported by the National Science Foundation through the Harvard University Materials Research Science and Engineering Center grant DMR-2011754.
