Proton Activity in Ionic Liquids at Elevated Temperatures
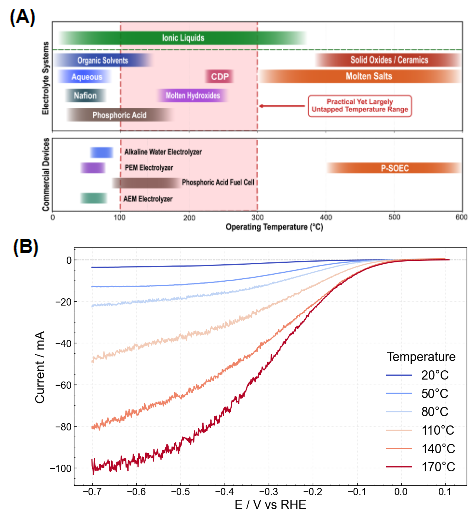
Electrochemical systems typically operate near ambient temperatures due to the volatility of aqueous electrolytes, limiting operations to around 80°C. However, recent work by Schiffer shows that electrochemical systems have better kinetics, and are potentially more energy efficient at intermediate temperatures (100-300°C). To reach these temperatures, we are developing ionic liquid (IL)-based electrochemical systems for efficient, modular chemical synthesis. Schiffer has been exploring diethylmethylammonium triflate ([Dema][TfO]) as a model protic IL. Based on preliminary data at temperatures up to 170°C, we have found that the prevailing theory identifying the most active proton is not complete. We are working to understanding the source of protons near an electrode at intermediate temperatures, and how proton activity can be controlled.
Zachary Schiffer (Applied Physics)
2025-2026 Harvard MRSEC (DMR-2011754)
